TRANSPARENCY: CoST Uganda Commissions 2nd Infrastructure Transparency Index
BY AWORI ESTHER LUCY
GENEVA|SHIFTMEDIA| There seems a ray of hope regarding a vaccine for the deadly Corona virus that has so far killed over 1.5 million people and infected over 54 million people worldwide.
The United States, India and Brazil top the globe with more infections and deaths due to Covid19
Uganda has so far registered over 16,000 cases and 145 deaths, while over 8,000 have recovered.
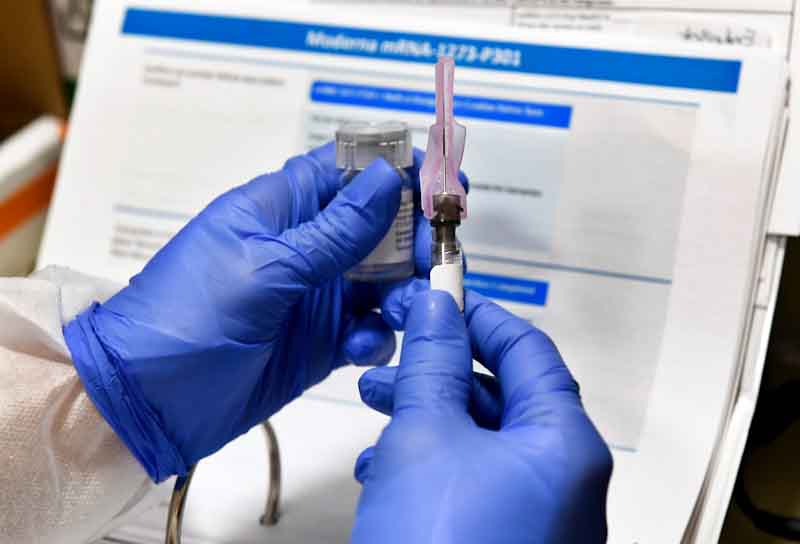
Now the good news is that Moderna Inc’s experimental COVID-19 vaccine has proved to be stable than expected at temperatures that ordinary refrigerators can provide and can be distributed using existing cold-chain shipping and storage infrastructure.
That opens the door for its early use in hard to reach places, such as Sub Saharan Africa that might not be appropriate for Pfizer Inc’s vaccine which needs ultra-cold storage.
The company said it expects the vaccine to be stable when kept at standard refrigerator temperatures of 2 to 8 degrees Celsius for 30 days, up from an initial projection of 7 days.
By comparison, Pfizer’s vaccine can be transported and stored for up to 6 months at minus 70 degrees Celsius (-94 F). It can be stored at standard refrigerator temperatures for up to five days.
We just announced that mRNA-1273, our COVID-19 vaccine candidate, has met its primary efficacy endpoint in the first interim analysis of the Phase 3 COVE study.
Read more: https://t.co/vYWEy8CKCv pic.twitter.com/YuLubU1tlx— Moderna (@moderna_tx) November 16, 2020
Shipping
Most Americans will be inoculated in May or June, according to Dr Anthony Fauci, the nation’s top infectious disease expert.
Under Operation Warp Speed’s distribution plan, the first group of shots will likely be distributed to and administered in closed settings, like hospitals and nursing homes.
After January, as supply increases, those shots will start to be available in pharmacies, doctors offices and clinics as well as mobile clinics, the plan shows.
Health officials have said it will be easier to use Moderna’s vaccine in these settings than Pfizer’s.